C12H22O11 11 H2SO4 12 C 11 H2SO4 11 H2O How do you know the concentrated sulfuric acid is a catalyst. If youre seeing this message it means were having trouble loading external resources on our website.

Double Replacement Reaction Definition And Examples
Predicting and balancing neutralization and precipitation reactions.
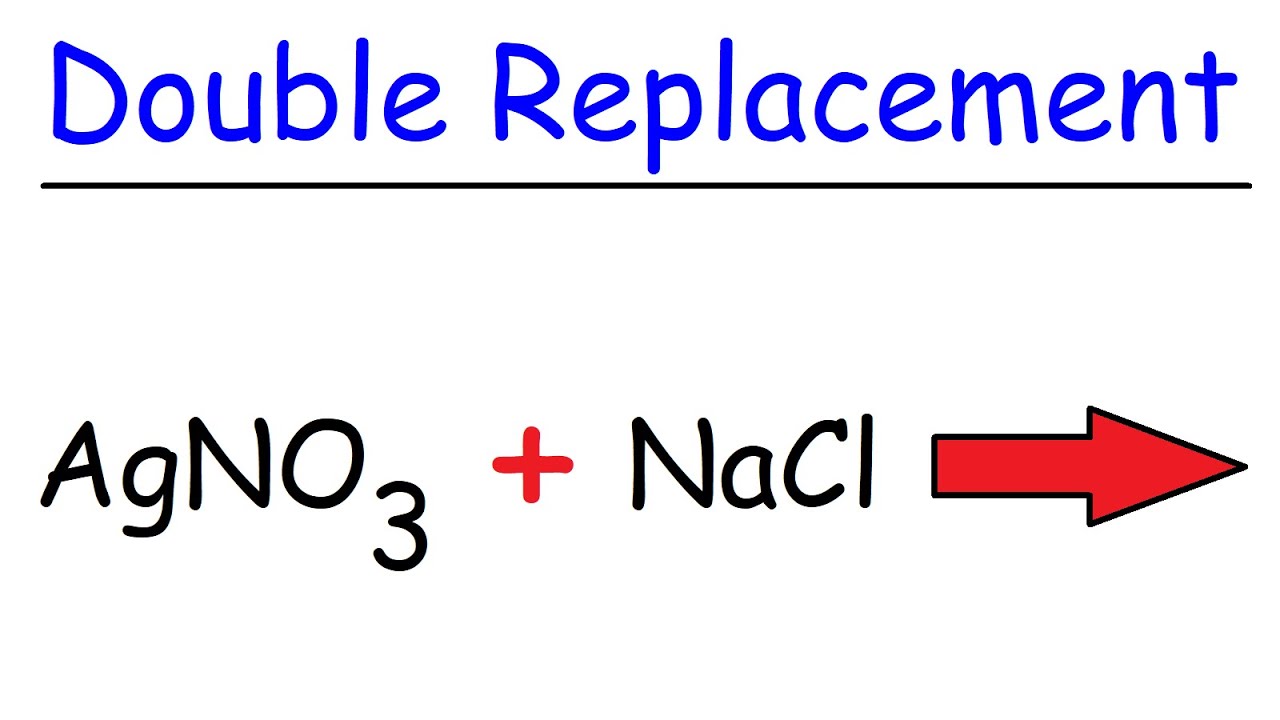
. Predicting and balancing neutralization and precipitation reactions. If youre behind a web filter. The sulfuric acid reacts with the sugar.
The sulfuric acid is not consumed or react with the reactant. A and B must be either different metals including H or different halogens. In a single-displacement reaction one element displaces another element in a compound.
CopperII chloride and leadII nitrate react in aqueous solutions by double replacement. What happens in a single displacement reaction. If 1027 g of copperII Chemistry.
The sulfuric acid forms new products. Write the balanced chemical equation the overall ionic equation and the net ionic equa- tion for this reaction. A B-C A-C B.
Definition and examples of double replacement reactions. Chemistry Chemical Reactions Single Replacement Reactions. The sulfuric acid is not consumed or react with the reactant.
Is the concentrated H2SO4 a catalyst in this reaction. Definition and examples of double replacement reactions. The general equation for a single-displacement reaction is.
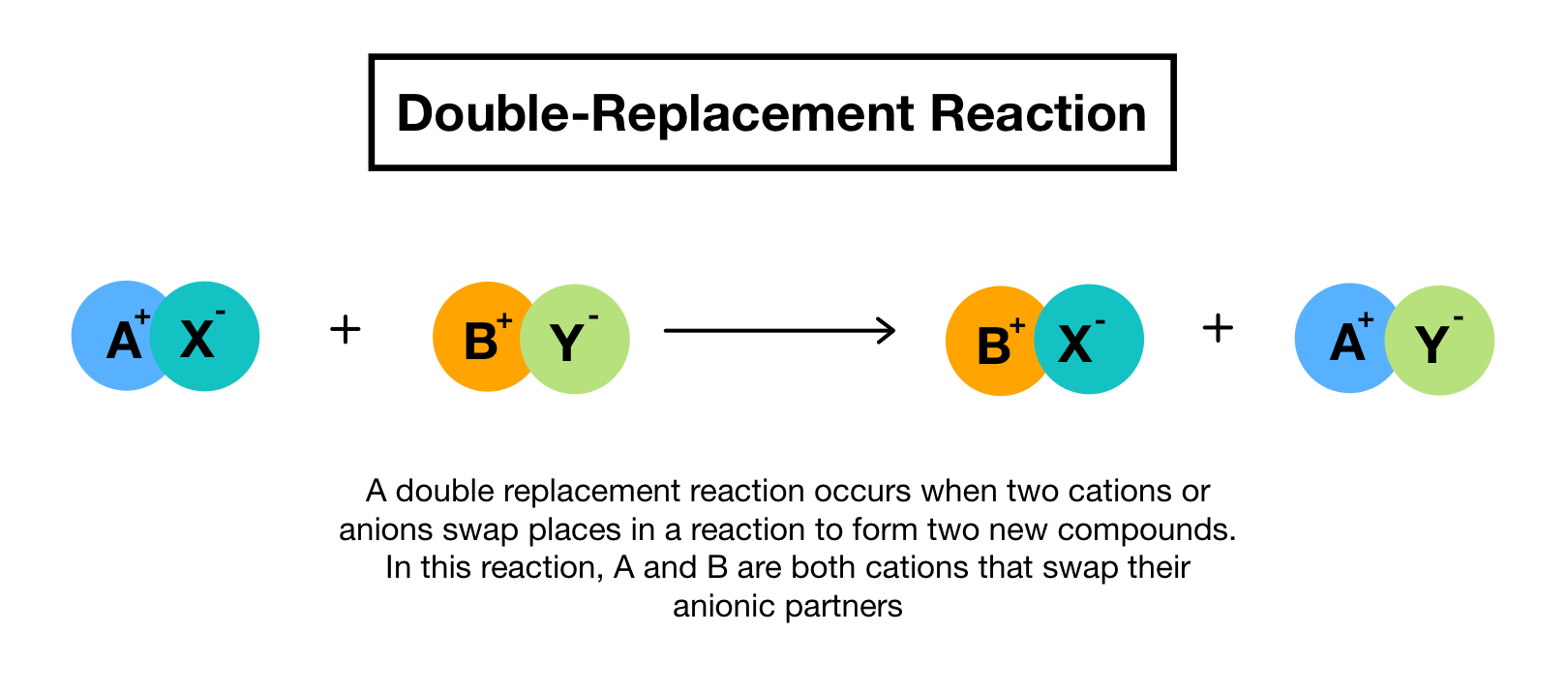
Double Replacement Reactions Definition Examples Expii
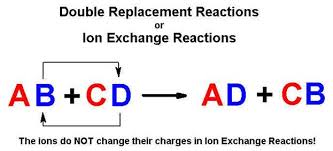
What Is A Double Replacement Reaction In Chemistry Socratic

Single Replacement Reaction Definition And Examples

Introduction To Double Replacement Reactions Youtube
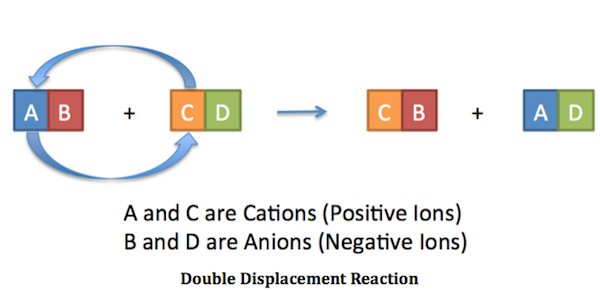
Double Replacement Reactions Chemistry Socratic
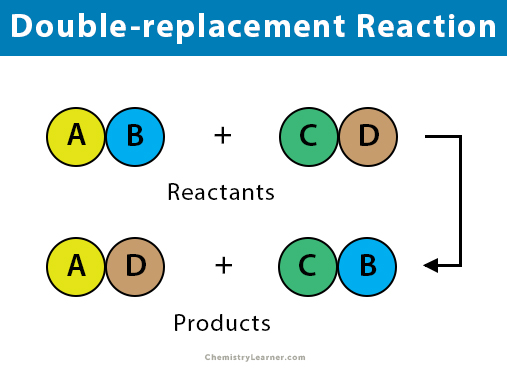
Double Replacement Double Displacement Reaction
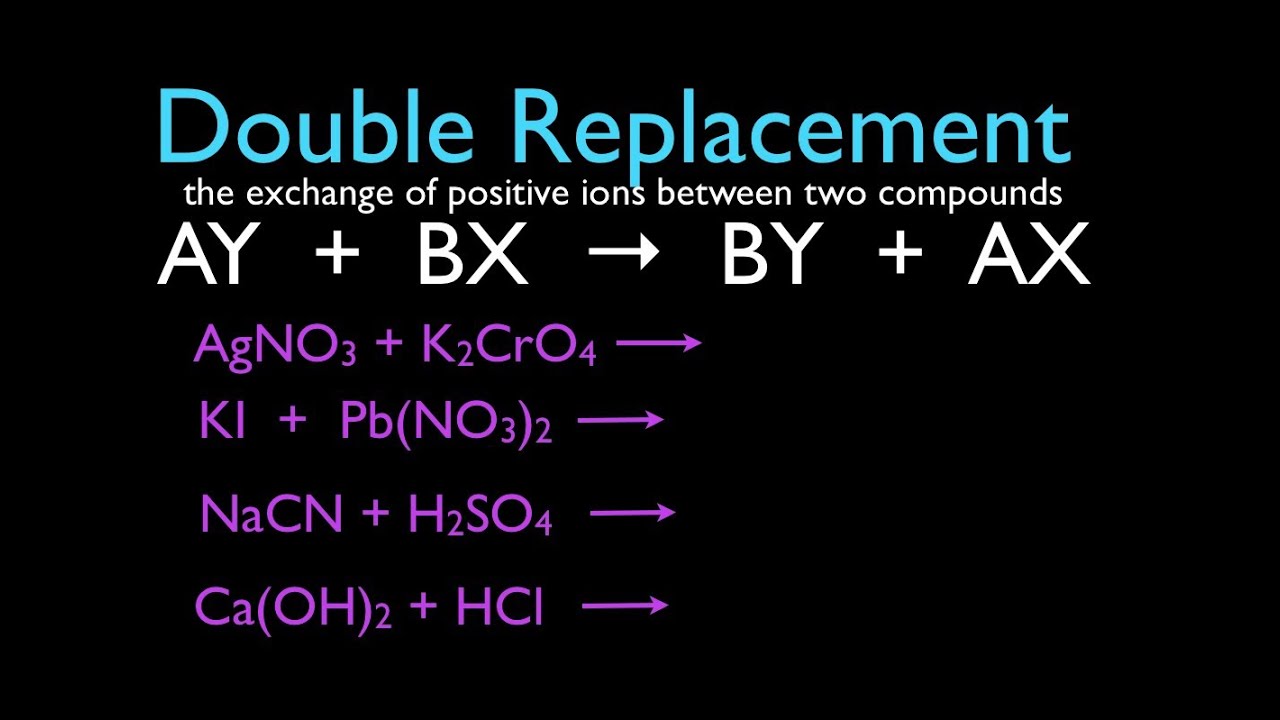
Chemical Reactions 1 Of 11 Double Replacement Reactions An Explanation Youtube

Chemical Reactions 2 Of 11 Single Replacement Reactions An Explanation Youtube
0 comments
Post a Comment